
LAST month, we talked about the importance of engaging in discussions with your Notified Body/Certification agency on transition planning for ISO 13485:2016 and the MDR.
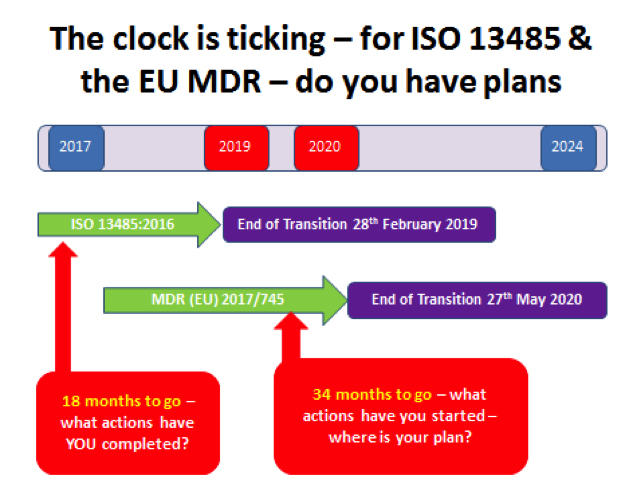
As you know, the ISO 13485:2016 was published in February 2016 and has a three-year transition period, due to end on February 28, 2019.
With only 18 months to go, you should be well on your way with your plans to transition from the previous version to the 2016 version, with particular attention being paid to existing certification expiration dates and audit cycle.
Discussions with your Notified Body are crucial, in order to ensure you understand their expectations and know what you need to do to maintain your certification.
You do not want to find that you have run out of time and that your certificate cannot be renewed because all the required actions have not been completed, potentially resulting in a certificate suspension – and the inevitable inability to ship your product.
Now, planning for the MDR, which also has a three year transition period, you should have started discussions with your Notified Body to begin to understand some of the challenges they need to work through.
Your regulatory leadership should be:
With such a large amount of work needing to be completed in a relatively short time, it is important that you plan and execute in a timely manner.
E-mail:
Tel: +44 (0) 141 946 6482
Address: Healthcare Skills Training International Ltd
West of Scotland Science Park
Block 7, Kelvin Campus
Glasgow G20 0SP